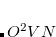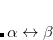9.7 A Brief Guide to Q-Chem’s Built-In ECPs
The remainder of this Chapter consists of a brief reference guide to Q-Chem’s built-in ECPs. The ECPs vary in their complexity and their accuracy and the purpose of the guide is to enable the user quickly and easily to decide which ECP to use in a planned calculation.
The following information is provided for each ECP:
The elements for which the ECP is available in Q-Chem. This is shown on a schematic Periodic Table by shading all the elements that are not supported.
The literature reference for each element for which the ECP is available in Q-Chem.
The matching orbital basis set that Q-Chem will use for light (i.e.. non-ECP atoms). For example, if the user requests SRSC ECPs—which are defined only for atoms beyond argon—Q-Chem will use the 6-311G* basis set for all atoms up to Ar.
The core electrons that are replaced by the ECP. For example, in the fit-LANL2DZ ECP for the Fe atom, the core is [Ne], indicating that the 1
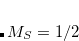 , 2
, 2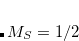 and 2
and 2 electrons are removed.
electrons are removed. The maximum spherical harmonic projection operator that is used for each element. This often, but not always, corresponds to the maximum orbital angular momentum of the core electrons that have been replaced by the ECP. For example, in the fit-LANL2DZ ECP for the Fe atom, the maximum projector is of
 -type.
-type. The number of valence basis functions of each angular momentum type that are present in the matching orbital basis set. For example, in the matching basis for the fit-LANL2DZ ECP for the Fe atom, there the three
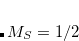 shells, three
shells, three  shells and two
shells and two  shells. This basis is therefore almost of triple-split valence quality.
shells. This basis is therefore almost of triple-split valence quality.
9.7.1 The fit-HWMB ECP at a Glance

(a) |
No ECP ; Pople STO-3G basis used |
(b) |
Wadt & Hay, J. Chem. Phys. 82 (1985) 285 |
(c) |
Hay & Wadt, J. Chem. Phys. 82 (1985) 299 |
(d) |
Hay & Wadt, J. Chem. Phys. 82 (1985) 270 |
Element |
Core |
Max Projector |
Valence |
H–He |
none |
none |
(1s) |
Li–Ne |
none |
none |
(2s,1p) |
Na–Ar |
[Ne] |
|
(1s,1p) |
K–Ca |
[Ne] |
|
(2s,1p) |
Sc–Cu |
[Ne] |
|
(2s,1p,1d) |
Zn |
[Ar] |
|
(1s,1p,1d) |
Ga–Kr |
[Ar]+3d |
|
(1s,1p) |
Rb–Sr |
[Ar]+3d |
|
(2s,1p) |
Y–Ag |
[Ar]+3d |
|
(2s,1p,1d) |
Cd |
[Kr] |
|
(1s,1p,1d) |
In–Xe |
[Kr]+4d |
|
(1s,1p) |
Cs–Ba |
[Kr]+4d |
|
(2s,1p) |
La |
[Kr]+4d |
|
(2s,1p,1d) |
Hf–Au |
[Kr]+4d+4f |
|
(2s,1p,1d) |
Hg |
[Xe]+4f |
|
(1s,1p,1d) |
Tl–Bi |
[Xe]+4f+5d |
|
(1s,1p) |
9.7.2 The fit-LANL2DZ ECP at a Glance

fit-LANL2DZ is not available for shaded elements
(a) |
No ECP ; Pople 6-31G basis used |
(b) |
Wadt & Hay, J. Chem. Phys. 82 (1985) 285 |
(c) |
Hay & Wadt, J. Chem. Phys. 82 (1985) 299 |
(d) |
Hay & Wadt, J. Chem. Phys. 82 (1985) 270 |
(e) |
Hay, J. Chem. Phys. 79 (1983) 5469 |
(f) |
Wadt, to be published |
Element |
Core |
Max Projector |
Valence |
H–He |
none |
none |
(2s) |
Li–Ne |
none |
none |
(3s,2p) |
Na–Ar |
[Ne] |
|
(2s,2p) |
K–Ca |
[Ne] |
|
(3s,3p) |
Sc–Cu |
[Ne] |
|
(3s,3p,2d) |
Zn |
[Ar] |
|
(2s,2p,2d) |
Ga–Kr |
[Ar]+3d |
|
(2s,2p) |
Rb–Sr |
[Ar]+3d |
|
(3s,3p) |
Y–Ag |
[Ar]+3d |
|
(3s,3p,2d) |
Cd |
[Kr] |
|
(2s,2p,2d) |
In–Xe |
[Kr]+4d |
|
(2s,2p) |
Cs–Ba |
[Kr]+4d |
|
(3s,3p) |
La |
[Kr]+4d |
|
(3s,3p,2d) |
Hf–Au |
[Kr]+4d+4f |
|
(3s,3p,2d) |
Hg |
[Xe]+4f |
|
(2s,2p,2d) |
Tl |
[Xe]+4f+5d |
|
(2s,2p,2d) |
Pb–Bi |
[Xe]+4f+5d |
|
(2s,2p) |
U–Pu |
[Xe]+4f+5d |
|
(3s,3p,2d,2f) |
Note that Q-Chem 4.2.2 and later versions also support LANL2DZ-SV basis, which employs SV basis functions (instead of 6-31G) on H, Li-He elements (like some other quantum chemistry packages).
9.7.3 The fit-SBKJC ECP at a Glance

(a) |
No ECP ; Pople 3-21G basis used |
(b) |
W.J. Stevens, H. Basch & M. Krauss, J. Chem. Phys. 81 (1984) 6026 |
(c) |
W.J. Stevens, M. Krauss, H. Basch & P.G. Jasien, Can. J. Chem 70 (1992) 612 |
(d) |
T.R. Cundari & W.J. Stevens, J. Chem. Phys. 98 (1993) 5555 |
Element |
Core |
Max Projector |
Valence |
H–He |
none |
none |
(2s) |
Li–Ne |
[He] |
|
(2s,2p) |
Na–Ar |
[Ne] |
|
(2s,2p) |
K–Ca |
[Ar] |
|
(2s,2p) |
Sc–Ga |
[Ne] |
|
(4s,4p,3d) |
Ge–Kr |
[Ar]+3d |
|
(2s,2p) |
Rb–Sr |
[Kr] |
|
(2s,2p) |
Y–In |
[Ar]+3d |
|
(4s,4p,3d) |
Sn–Xe |
[Kr]+4d |
|
(2s,2p) |
Cs–Ba |
[Xe] |
|
(2s,2p) |
La |
[Kr]+4d |
|
(4s,4p,3d) |
Ce–Lu |
[Kr]+4d |
|
(4s,4p,1d,1f) |
Hf–Tl |
[Kr]+4d+4f |
|
(4s,4p,3d) |
Pb–Rn |
[Xe]+4f+5d |
|
(2s,2p) |
9.7.4 The fit-CRENBS ECP at a Glance

fit-CRENBS is not available for shaded elements
(a) |
No ECP ; Pople STO-3G basis used |
(b) |
Hurley, Pacios, Christiansen, Ross & Ermler, J. Chem. Phys. 84 (1986) 6840 |
(c) |
LaJohn, Christiansen, Ross, Atashroo & Ermler, J. Chem. Phys. 87 (1987) 2812 |
(d) |
Ross, Powers, Atashroo, Ermler, LaJohn & Christiansen, J. Chem. Phys. 93 (1990) 6654 |
Element |
Core |
Max Projector |
Valence |
H–He |
none |
none |
(1s) |
Li–Ne |
none |
none |
(2s,1p) |
Na–Ar |
none |
none |
(3s,2p) |
K–Ca |
none |
none |
(4s,3p) |
Sc–Zn |
[Ar] |
|
(1s,0p,1d) |
Ga–Kr |
[Ar]+3d |
|
(1s,1p) |
Y–Cd |
[Kr] |
|
(1s,1p,1d) |
In–Xe |
[Kr]+4d |
|
(1s,1p) |
La |
[Xe] |
|
(1s,1p,1d) |
Hf–Hg |
[Xe]+4f |
|
(1s,1p,1d) |
Tl–Rn |
[Xe]+4f+5d |
|
(1s,1p) |
9.7.5 The fit-CRENBL ECP at a Glance

(a) |
No ECP ; Pople 6-311G* basis used |
(b) |
Pacios & Christiansen, J. Chem. Phys. 82 (1985) 2664 |
(c) |
Hurley, Pacios, Christiansen, Ross & Ermler, J. Chem. Phys. 84 (1986) 6840 |
(d) |
LaJohn, Christiansen, Ross, Atashroo & Ermler, J. Chem. Phys. 87 (1987) 2812 |
(e) |
Ross, Powers, Atashroo, Ermler, LaJohn & Christiansen, J. Chem. Phys. 93 (1990) 6654 |
(f) |
Ermler, Ross & Christiansen, Int. J. Quantum Chem. 40 (1991) 829 |
(g) |
Ross, Gayen & Ermler, J. Chem. Phys. 100 (1994) 8145 |
(h) |
Nash, Bursten & Ermler, J. Chem. Phys. 106 (1997) 5133 |
Element |
Core |
Max Projector |
Valence |
H–He |
none |
none |
(3s) |
Li–Ne |
[He] |
S |
(4s,4p) |
Na–Mg |
[He] |
S |
(6s,4p) |
Al–Ar |
[Ne] |
P |
(4s,4p) |
K–Ca |
[Ne] |
P |
(5s,4p) |
Sc–Zn |
[Ne] |
P |
(7s,6p,6d) |
Ga–Kr |
[Ar] |
P |
(3s,3p,4d) |
Rb–Sr |
[Ar]+3d |
D |
(5s,5p) |
Y–Cd |
[Ar]+3d |
D |
(5s,5p,4d) |
In–Xe |
[Kr] |
D |
(3s,3p,4d) |
Cs–La |
[Kr]+4d |
D |
(5s,5p,4d) |
Ce–Lu |
[Xe] |
D |
(6s,6p,6d,6f) |
Hf–Hg |
[Kr]+4d+4f |
F |
(5s,5p,4d) |
Tl–Rn |
[Xe]+4f |
F |
(3s,3p,4d) |
Fr–Ra |
[Xe]+4f+5d |
F |
(5s,5p,4d) |
Ac–Pu |
[Xe]+4f+5d |
F |
(5s,5p,4d,4f) |
Am–Lr |
[Xe]+4f+5d |
F |
(0s,2p,6d,5f) |
9.7.6 The SRLC ECP at a Glance

(a) |
No ECP ; Pople 6-31G basis used |
(b) |
Fuentealba, Preuss, Stoll & Szentpaly, Chem. Phys. Lett. 89 (1982) 418 |
(c) |
Fuentealba, Szentpály, Preuss & Stoll, J. Phys. B 18 (1985) 1287 |
(d) |
Bergner, Dolg, Küchle, Stoll & Preuss, Mol. Phys. 80 (1993) 1431 |
(e) |
Nicklass, Dolg, Stoll & Preuss, J. Chem. Phys. 102 (1995) 8942 |
(f) |
Schautz, Flad & Dolg, Theor. Chem. Acc. 99 (1998) 231 |
(g) |
Fuentealba, Stoll, Szentpaly, Schwerdtfeger & Preuss, J. Phys. B 16 (1983) L323 |
(h) |
Szentpaly, Fuentealba, Preuss & Stoll, Chem. Phys. Lett. 93 (1982) 555 |
(i) |
Küchle, Dolg, Stoll & Preuss, Mol. Phys. 74 (1991) 1245 |
(j) |
Küchle, to be published |
Element |
Core |
Max Projector |
Valence |
H–He |
none |
none |
(2s) |
Li–Be |
[He] |
|
(2s,2p) |
B–N |
[He] |
|
(2s,2p) |
O–F |
[He] |
|
(2s,3p) |
Ne |
[He] |
|
(4s,4p,3d,1f) |
Na–P |
[Ne] |
|
(2s,2p) |
S–Cl |
[Ne] |
|
(2s,3p) |
Ar |
[Ne] |
|
(4s,4p,3d,1f) |
K–Ca |
[Ar] |
|
(2s,2p) |
Zn |
[Ar]+3d |
|
(3s,2p) |
Ga–As |
[Ar]+3d |
|
(2s,2p) |
Se–Br |
[Ar]+3d |
|
(2s,3p) |
Kr |
[Ar]+3d |
|
(4s,4p,3d,1f) |
Rb–Sr |
[Kr] |
|
(2s,2p) |
In–Sb |
[Kr]+4d |
|
(2s,2p) |
Te–I |
[Kr]+4d |
|
(2s,3p) |
Xe |
[Kr]+4d |
|
(4s,4p,3d,1f) |
Cs–Ba |
[Xe] |
|
(2s,2p) |
Hg–Bi |
[Xe]+4f+5d |
|
(2s,2p,1d) |
Po–At |
[Xe]+4f+5d |
|
(2s,3p,1d) |
Rn |
[Xe]+4f+5d |
|
(2s,2p,1d) |
Ac–Lr |
[Xe]+4f+5d |
|
(5s,5p,4d,3f,2g) |
9.7.7 The SRSC ECP at a Glance

SRSC is not available for shaded elements
(a) |
No ECP ; Pople 6-311G* basis used |
(b) |
Leininger, Nicklass, Küchle, Stoll, Dolg & Bergner, Chem. Phys. Lett. 255 (1996) 274 |
(c) |
Kaupp, Schleyer, Stoll & Preuss, J. Chem. Phys. 94 (1991) 1360 |
(d) |
Dolg, Wedig, Stoll & Preuss, J. Chem. Phys. 86 (1987) 866 |
(e) |
Andrae, Haeussermann, Dolg, Stoll & Preuss, Theor. Chim. Acta 77 (1990) 123 |
(f) |
Dolg, Stoll & Preuss, J. Chem. Phys. 90 (1989) 1730 |
(g) |
Küchle, Dolg, Stoll & Preuss, J. Chem. Phys. 100 (1994) 7535 |
Element |
Core |
Max Projector |
Valence |
H–Ar |
none |
none |
(3s) |
Li–Ne |
none |
none |
(4s,3p,1d) |
Na–Ar |
none |
none |
(6s,5p,1d) |
K |
[Ne] |
|
(5s,4p) |
Ca |
[Ne] |
|
(4s,4p,2d) |
Sc–Zn |
[Ne] |
|
(6s,5p,3d) |
Rb |
[Ar]+3d |
|
(5s,4p) |
Sr |
[Ar]+3d |
|
(4s,4p,2d) |
Y–Cd |
[Ar]+3d |
|
(6s,5p,3d) |
Cs |
[Kr]+4d |
|
(5s,4p) |
Ba |
[Kr]+4d |
|
(3s,3p,2d,1f) |
Ce–Yb |
[Ar]+3d |
|
(5s,5p,4d,3f) |
Hf–Pt |
[Kr]+4d+4f |
|
(6s,5p,3d) |
Au |
[Kr]+4d+4f |
|
(7s,3p,4d) |
Hg |
[Kr]+4d+4f |
|
(6s,6p,4d) |
Ac–Lr |
[Kr]+4d+4f |
|
(8s,7p,6d,4f) |
9.7.8 The Karlsruhe “def2” ECP at a Glance
For elements Rb–Rn (not including the lanthanides), all the Karlsruhe “def2" basis sets are paired with a common set of ECPs.[Weigend and Ahlrichs(2005)] It is briefly summarized in the table below (the number of valence basis functions depend on the basis set in use so it is not presented):
Element |
Core |
Max Projector |
H–Kr |
none |
none |
Rb–Xe |
[Ar]+3d |
|
Cs–La |
[Kr]+4d |
|
Hf–Rn |
[Kr]+4d+4f |
|